Overview:
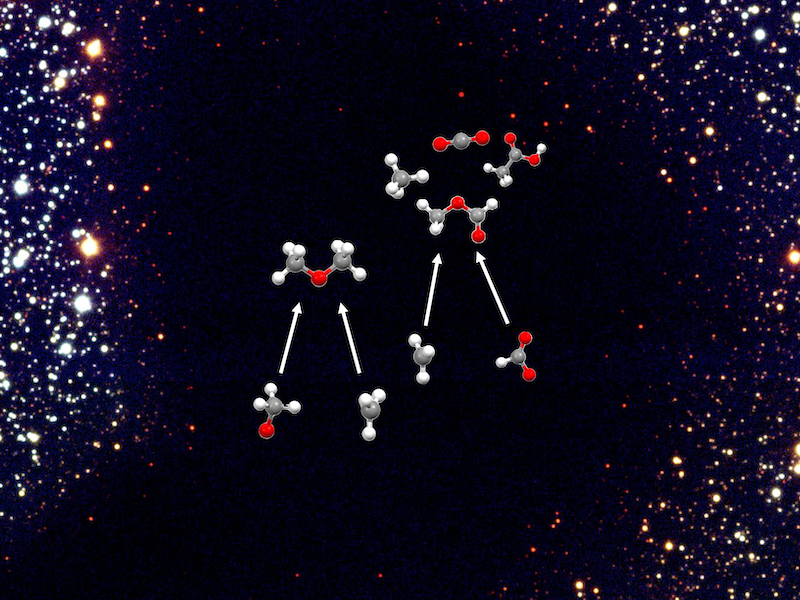
Project Researcher at the Astrobiology Center Yuu Komatsu and Project Assistant Professor at National Astronomical Observatory of Japan Kenji Furukawa used the automated reaction path exploration method based on quantum chemistry to investigate the process by which representative complex organic molecules detected in interstellar space, such as dimethyl ether and methyl formate, are formed. As a result, they discovered pathways through which reactions can proceed for each molecule within extremely cold (~10K) molecular clouds (see Figure 1). This study applies the numerical simulations, aiming to explore energetically feasible reaction pathways, based on theoretical chemistry to astronomy and contributes to understanding what occurs behind the observations of complex organic matter. This research achievement was published online in the journal “ACS Earth and Space Chemistry” on August 17, 2023.
Research Context:
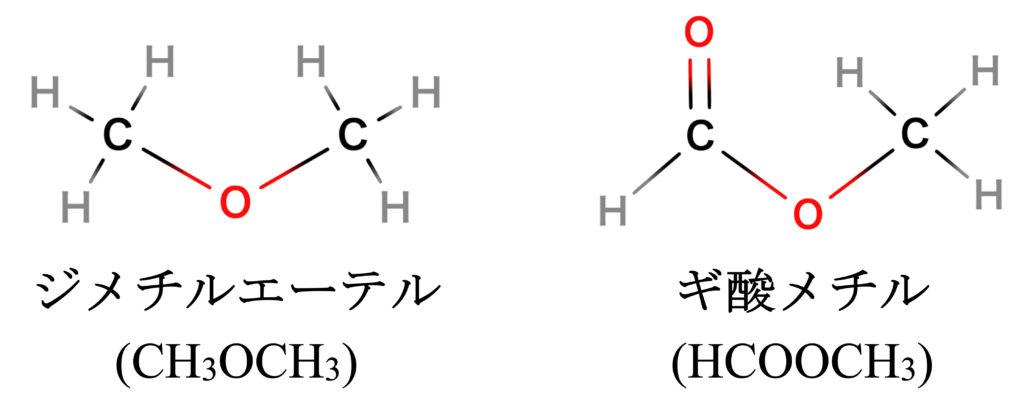
High-temperature regions reaching >100 K (Note 1) exist in massive and small-mass protostar forming regions, and a wide variety of complex organic molecules have been detected in these regions. Dimethyl ether and methyl formate (Figure 2) have been typically observed in molecular cloud cores where stars are forming, and their formation has been thought to be mainly due to chemical reactions in the hot (>100 K) gas phase after star formation or radical chemical reactions on the warm dust surface (>20 K). Recently, however, these molecules have been observed in molecular cloud cores where stars have not yet formed, even at cryogenic temperatures as low as 10 K. This has necessitated a reexamination of how these molecules are formed. For dimethyl ether, formation by radiative bonding in the gas phase (Note 2) has been considered, but has yet to explain the amounts observed in low-temperature environments. In addition, the formation process of methyl formate has not been well studied in the first place.
Research Findings:
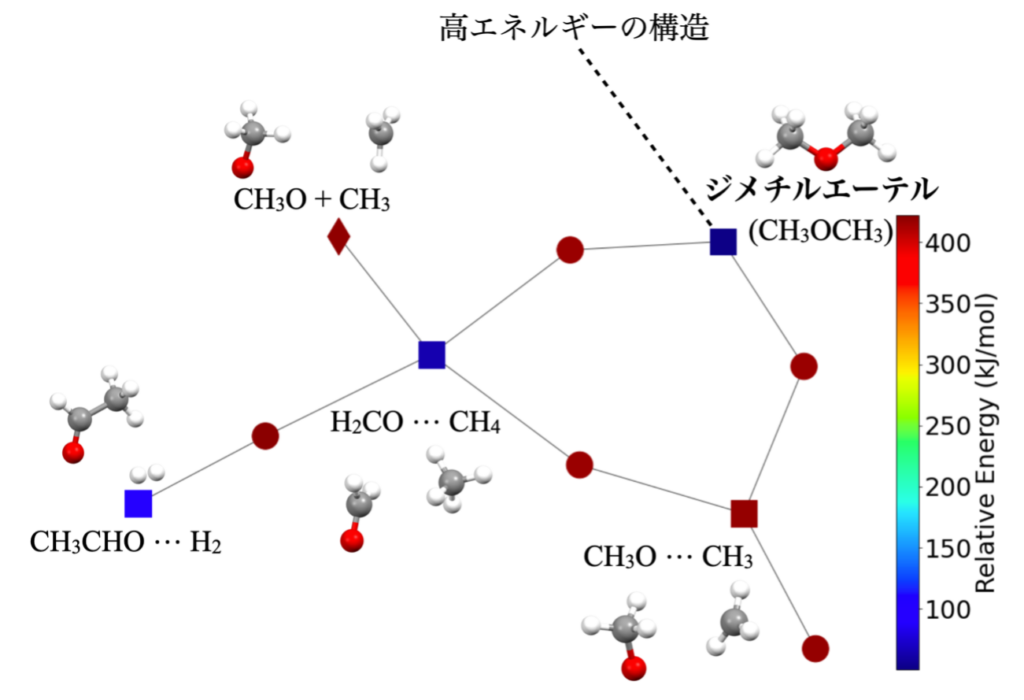
In order to clarify the formation process of dimethyl ether and methyl formate at cryogenic temperatures, an automated chemical reaction pathway pathway search method based on the transition state theory (Note 4) of quantum chemistry (Note 3) (the same method was used in Komatsu and Suzuki, ACS Earth Space Chem. (the same method used in Komatsu and Suzuki, ACS Earth Space Chem. 2022) was used to investigate the pathways by which these molecules can be energetically produced in their electronic ground state (Note 5). The method used here is to complete the energy profile of each possible conformation for each of these target molecules, and when a target molecule splits into two molecules, the pathway to the target molecule is picked up in reverse order to extract the pathway that requires less external energy. This method was adopted.
The calculations revealed that for both molecules, the formation pathway is a gas-phase exothermic reaction with no reaction barriers. For dimethyl ether, the obtained reaction network (Figure 3) revealed a formation pathway from CH3O and CH3. This was partially inferred from previous studies, and a more comprehensive pathway consistent with these was obtained. On the other hand, a more complex formation pathway was inferred for methyl formate. A pathway that proceeds without reaction barriers was found, but the main products are carbon dioxide and methane, and methyl formate was found to be only a by-product. For methyl formate, not only gas-phase reactions but also other reaction pathways, such as dust surface reactions, may be more important. Thus, this study theoretically predicts the pathways by which these complex organic molecules can be formed even at cryogenic temperatures, and will provide fundamental guidance in elucidating the full picture of how complex organic molecules are formed.
In the future, it will be important to connect the obtained promising reaction networks with models based on reaction rate equations to estimate the amount of complex organic molecules in interstellar space from the viewpoint of comparing theory and observation. In addition, although the present calculations were limited to gas-phase reactions, a similar study of dust surface reactions would be useful as an evaluation using quantum chemical calculations.
Annotation:
(1) 0 ºC is 273.15 K. 100 K is -173.15 ºC.
2) A phenomenon in which atoms and molecules collide in the gas phase, radiating electromagnetic waves to form stable molecules. In English, radiative association.
(3) This is a field in which quantum mechanics is applied to various problems in chemistry. By solving the wave equation of a system using quantum chemical calculations, the physical properties and reactivity of the system can be investigated.
(4) Transition states, which are intermediate structures between reactants and products, are identified and their energies are evaluated to estimate the ease with which a reaction can proceed.
(5) The electron configuration with the lowest energy in an atomic molecule. For example, when light from a star acts on a molecule, it enters an electronically excited state with high energy. This study does not deal with this excited state.
Publication:
Journal:ACS Earth and Space Chemistry
“The Automated Reaction Pathway Search Reveals the Energetically Favorable Synthesis of Interstellar CH3OCH3 and HCOOCH3“
Authors:Isamu Komatsu(1),(2), Kenji Furuya(2)
(1) Astrobiology Center, (2) National Astronomical Observatory of Japan
DOI:10.1021/acsearthspacechem.3c00117
Paper Links:https://pubs.acs.org/doi/10.1021/acsearthspacechem.3c00117
Related Links
National Astronomical Observatory of Japan Scientific Research Division Press Release