Key Points:
- Plants and algae typically utilize only visible light for photosynthesis in the sunlight spectrum. Certain algae species thriving in Antarctica can harness infrared light for photosynthesis, but the mechanism was previously unknown.
- Using a device called a cryogenic electron microscope, they revealed the protein structure these algae utilize to conduct photosynthesis with infrared light.
- Many planets discovered outside the solar system orbit stars which have so lower-temperature than the Sun that they emit primarily infrared light. This suggests the potential for life that utilizes infrared light for photosynthesis. This achievement may provide insights into exploring such possibilities of life beyond Earth.
Research Overview:
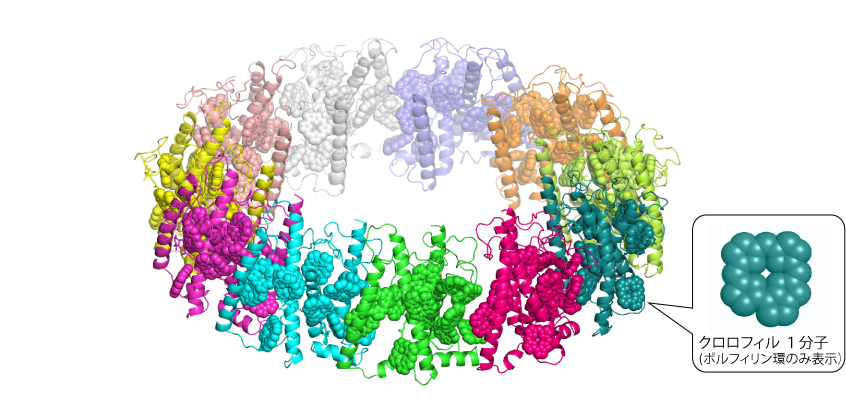
A research team led by Dr. Makiko Kosugi from the Astrobiology Center (currently a Project Assistant Professor at the National Institute of Basic Biology and a Collaborative Researcher at Chuo University), along with researchers from Institute of Materials Structure Science at the High Energy Accelerator Research Organization (KEK) including Associate Professor Masato Kawasaki, Project Assistant Professors Naruhiko Adachi and Toshio Moriya, and Professor Toshiya Chida, from Tohoku University Associate Professor Minoru Shibata, from Akita Prefectural University Associate Professor Kojiro Hara, from Tokyo University of Agriculture Professor Shinichi Takaichi, from the National Institute of Basic Biology RMC Professor Yasuhiro Kamei, from University of Hyogo Associate Professor Yasuhiro Kashino , from the National Institute of Polar Research Professor Sakae Kudo, and from Chuo University Professor Hiroyuki Oda, identified the light-harvesting antenna protein (Pc-frLHC) responsible for absorbing far-red light (700-800 nm) in the green algae Prasiola crispa, which is known to conduct oxygenic photosynthesis utilizing far-red light, part of infrared light. The team also revealed the three-dimensional structure of this molecule by Single Particle Analysis (Note 1) using a cryogenic electron microscope at KEK, revealing that Pc-frLHC forms a large complex composed of 11 identical proteins in a ring structure (see Figure 1). Each protein binds 11 chlorophyll molecules, with five of these chlorophylls suggested to be special for absorbing far-red light. Spectroscopic analysis indicated that part of the far-red light energy absorbed by these special chlorophylls is converted within Pc-frLHC to energy equivalent to visible light, which is then utilized for photosynthesis. This result was published in the British scientific journal “Nature Communications” on February 15, 2023 (Kosugi et al., 2023, “Uphill energy transfer mechanism for photosynthesis in an Antarctic alga”).
Research Background:
Photosynthesis by plants and algae uses the energy of visible light (350-700 nm) in sunlight to break down water into oxygen, hydrogen, and electrons, and the reducing power obtained is used for carbon dioxide assimilation. It is usually not used for water splitting because its energy is lower than that of visible light. Although some cyanobacteria (see Note 2) have been known to utilize infrared light for photosynthesis and have been analyzed, eukaryotic photosynthetic organisms such as plants and algae (see Note 2) have not been analyzed.
Kosugi et al. at the Center for Astrobiology (now the National Institute for Basic Biology) and colleagues have recently shown that the green alga Nankyoku Kawanori, a eukaryotic photosynthetic organism that grows on land in Antarctica, uses far-red light (700~800 nm), which is part of infrared light, for photosynthesis with an energy conversion efficiency as high as that of visible light (Kosugi (Kosugi et al. 2020). The terrestrial environment in Antarctica is extremely dry with low temperatures and frequent freezing. In addition, the extremely strong ultraviolet rays during the summer make it impossible for many organisms to survive. Nankyoku Kawanori is extremely resistant to desiccation and freezing, and can quickly regain its metabolic activity after being dried out or frozen for a long period of time by applying water. These characteristics make it one of the few photosynthetic organisms that can grow in the Antarctic terrestrial environment. Nankyo kawanori forms colonies (aggregates) with many layers of cells (Fig. 2).

Near the surface of the colony, sunlight can reach enough to use visible light for photosynthesis, but this has the disadvantage of damaging the cells due to the ultraviolet rays contained in the sunlight. On the other hand, the lower layers of the colony are less likely to be damaged by ultraviolet light, but visible light is absorbed by the algae on the surface layer and used for photosynthesis, so very little visible light reaches the colony. It is thought that Nankei-Kawanori acquired a system that utilizes infrared light for photosynthesis during its evolution and increased the amount of photosynthesis in the lower layers of its colony, allowing it to reproduce even in the extremely harsh environment of Antarctica (Fig. 3).
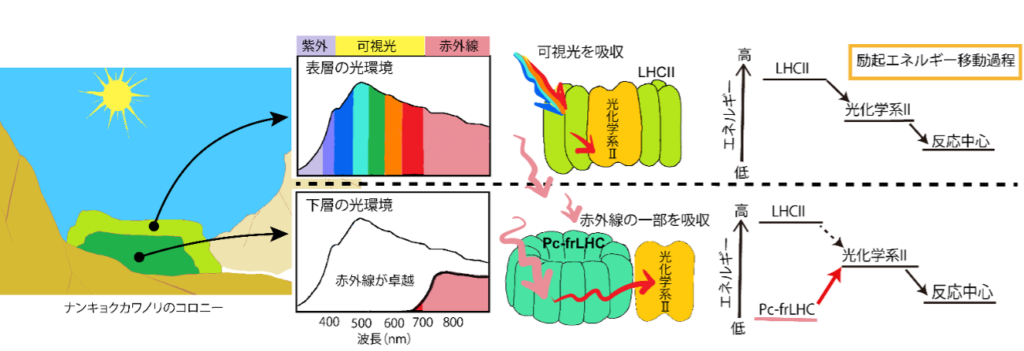
The existence of organisms that can use infrared light for photosynthesis has attracted much attention in the field of astrobiology. This is because many of the exoplanets discovered so far are located around low-temperature stars that are fainter than the sun and emit more infrared light than visible light (Note 3). Oxygen emitted into the atmosphere by photosynthetic organisms is considered to be one of the traces of life that can be observed from Earth when studying the presence of life on exoplanets. Elucidating the mechanism and evolutionary process of photosynthesis using infrared light on Earth is important for discussing the possibility of detecting oxygen on exoplanets around low-temperature stars. It has been suggested that infrared-enabled photosynthesis in Nankyoku Kawanori involves an uphill energy transfer (Note 4), in which low energy excites molecules at high energy levels, and the mechanism that achieves high light-use efficiency may involve a previously unknown quantum biological reaction The mechanism of high light-utilization efficiency may involve a previously unknown quantum biological reaction. Therefore, we purified and identified an infrared harvesting antenna protein from Nankyo kawanori and elucidated its molecular structure to elucidate the mechanism of infrared-utilizing photosynthesis.
Research Findings:
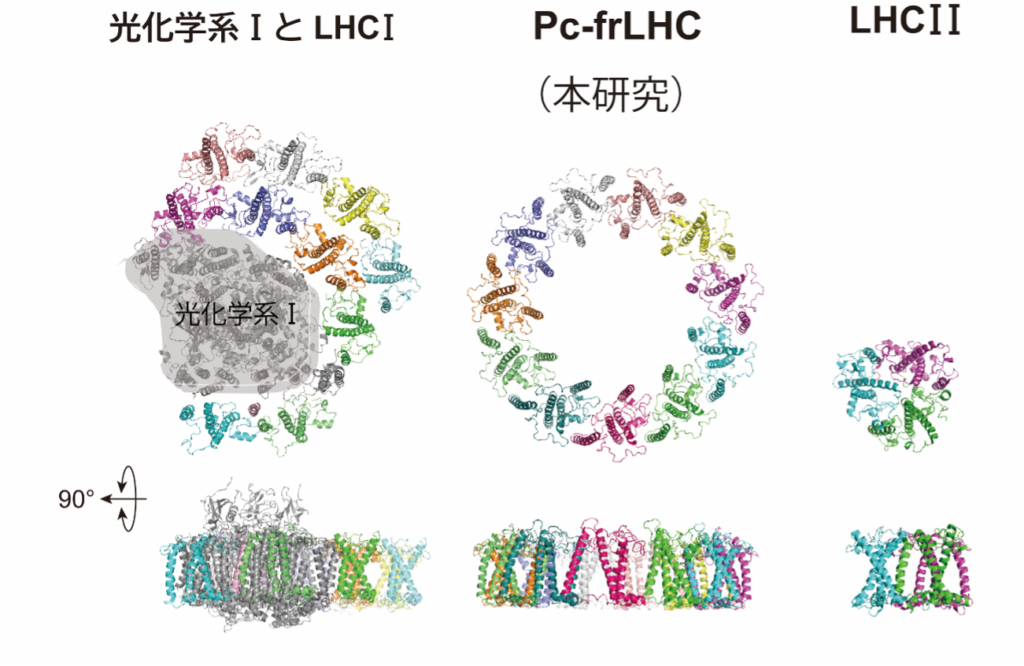
The Nankyoku Kawanori used in the experiment were collected during the activities of the 49th and 54th Expeditions to the Antarctic region. The cells of the Nankyoku Kawanori were crushed and separated according to protein size and charge, and a protein with a pronounced absorption band in far-red light was purified and named Pc-frLHC (Prasiola crispa far-red light harvesting Chl-binding protein The protein was named Pc-frLHC (Prasiola crispa far-red light harvesting Chl-binding protein complex). Analysis of the amino acid sequence of the protein revealed that Pc-frLHC is a four-transmembrane (Note 6) LHCI (Light harvesting chlorophyll a/b binding complex of photosystem I), which binds to photosystem I (Note 5) in some green algae. This four-transmembrane LHCI has been reported to absorb the longest wavelength of visible light in the green alga Chlamydomonas, but can hardly absorb far-red light (Mozzo et al. 2010). Furthermore, Pc-frLHC functions as an antenna for photosystem II (Note 5), which performs water splitting, rather than photosystem I, suggesting that the absorption band of the long-wavelength absorbing LHC originally possessed by green algae moved to an even longer wavelength and evolved as an antenna for photosystem II.
Single-particle analysis using cryo-electron microscopy has succeeded in obtaining a three-dimensional structural molecular model of Pc-frLHC with high resolution. The general photosystem II antenna protein of green algae is a structure of three proteins bound together, but the Pc-frLHC analyzed in this study is a novel complex structure with 11 proteins bound together in a ring (Fig. 4). 11 chlorophylls are bound to one protein, and all of them in the ring chlorophylls are located at a distance that allows energy transfer, forming an energetically connected network. Chlorophyll normally absorbs visible light, but it is known that when multiple chlorophyll molecules interact with each other in close proximity, a portion of the absorption band shifts to the longer wavelength side. In Chlamydomonas quadruple transmembrane LHCI, which can absorb relatively long-wavelength light, two chlorophylls were reported to be in close proximity (Mozzo et al. 2010), but in Pc-frLHC, these two chlorophylls were found to be in close proximity to another chlorophyll, indicating that five chlorophylls strongly interact. This chlorophyll structure is the basis of the Pc-frLHC. This chlorophyll structure is thought to be responsible for the far-red light absorption of Pc-frLHCs.
To understand how far-red light energy absorbed by Pc-frLHC is transferred, we excited the long-wavelength absorbing chlorophyll of Pc-frLHC with ultrashort laser pulses of far-red light and examined how chlorophyll fluorescence (see Note 7) changes over time. The fluorescence from the long-wavelength-absorbing chlorophyll is detected at 713 nm, while the fluorescence from normal chlorophyll is detected at 680 nm; by examining how the fluorescence at 680 nm increases with time, we can see that the energy between the long-wavelength-absorbing and normal chlorophylls goes within 25 picoseconds (= 0. It has been found that the energy goes back and forth within 25 picoseconds (= 0.000000025 seconds). This result indicates that an uphill excitation energy transfer from long-wavelength-absorbed chlorophyll to normal chlorophyll is indeed occurring within Pc-frLHC. In this process, part of the energy of far-red light is converted to visible light energy, and the subsequent photosynthetic reaction is thought to proceed in the same way as when visible light is absorbed.
Perspectives:
- Elucidating the Details of Uphill Excitation Energy Transfer
In order to elucidate the full extent of the high excitation efficiency of photosystem II by far-red light, it is necessary to analyze the details of the energy transfer from Pc-frLHC to photosystem II. To this end, we will purify a supercomplex of Pc-frLHC and photosystem II from Nankei-kawanori cells to elucidate the excitation energy transfer process - Evolutionary aspects of far-red light-utilizing oxygen-evolving photosynthetic organisms
Although the protein has not been identified or structurally analyzed, several eukaryotic algae with a prominent far-red light absorption band have been reported in addition to Nankyoku Kawanori, and it is possible that a far-red light-absorbing light-harvesting protein similar to the Pc-frLHC found in Nankyoku Kawanori in this study exists in other eukaryotic algae as well Pc-frLHC found in Nankyokawanori. We will obtain amino acid sequences of far-red light-utilizing light-harvesting proteins in various algae, clarify their evolutionary lineages, and analyze the homology and diversity of far-red light-utilizing mechanisms. - Astrobiological Aspects
The search for life targeting exoplanets is expected to make great progress in the future with the development of the next-generation Very Large Telescope. Oxygen is a promising bio-signature, but is there any possibility of detecting “photosynthetically derived” oxygen on exoplanets around low-temperature stars? By clarifying the details of infrared oxygen-evolving photosynthesis on Earth, we will explore the possibility of the evolution of photosynthetic life on exoplanets around low-temperature stars.
Annotation:
1) Single particle analysis by cryo-electron microscopy:
Protein structure analysis technology has developed rapidly in recent years. Conventional X-ray protein structure analysis requires protein crystals, but cryo-electron microscopy eliminates the need for crystals, facilitating analysis of samples that are difficult to crystallize, or samples that can only be obtained in small quantities, such as the Antarctic-derived organisms in this study.
2) Cyanobacteria and eukaryotic photosynthetic organisms:
Cyanobacteria are the most primitive oxygen-evolving photosynthetic organisms and are considered to be the ancestors of chloroplasts. Cyanobacteria evolved through intracellular symbiosis into eukaryotic photosynthetic organisms such as algae and plants. The mechanism of photosynthesis by far-red light is different between cyanobacteria and eukaryotic photosynthetic organisms, and it is important to clarify both mechanisms.
3) Low-temperature stars:
A star that is lighter and cooler than the Sun (G-type), also known as an M-type dwarf star. They are also called M-type dwarfs. They are considered important targets in the search for life because of their overwhelmingly large percentage of the stars in the Universe. Because the ratio of infrared light is higher than that of visible light, the environment of the surrounding exoplanets is also dominated by infrared light.
4) Uphill excitation energy transfer:
Excitation energy transfer between chlorophyll molecules is usually passed from a higher energy level molecule to a lower energy level molecule, and this reverse reaction is called uphill excitation energy transfer. Uphill excitation energy transfer is considered to occur when the energy difference between molecules is compensated by thermal energy.
5) Photosystem I and Photosystem II:
Proteins involved in the electron transfer system in the thylakoid membrane of chloroplasts. It has a special chlorophyll reaction center that causes charge separation by light energy. Photosystem II breaks down water, and photosystem I increases the energy level of electrons received from photosystem II to a level that allows the reduction of electron transfers needed for carbon dioxide fixation. Excitation of photosystem II requires higher light energy at shorter wavelengths than that of photosystem I.
6) Four transmembrane LHCs:
Most light-harvesting antenna proteins in algae are folded and embedded in a lipid bilayer called the thylakoid membrane in chloroplasts. The number of times they penetrate the membrane depends on the number of folds.
7) Chlorophyll fluorescence:
Light emitted when chlorophyll is electronically excited by light to a lower energy state.
論文情報:
Journal:Nature Communications
“Uphill energy transfer mechanism for photosynthesis in an Antarctic alga”
Author: Makiko Kosugi, Masato Kawasaki, Yutaka Shibata, Kojiro Hara, Shinichi Takaichi, Toshio Moriya, Naruhiko Adachi, Yasuhiro Kamei, Yasuhiro Sagano, Sakae Kudo, Hiroyuki Koike, Toshiya Senda
DOI: 10.1038/s41467-023-36245-1
URL:https://www.nature.com/articles/s41467-023-36245-1
Co-Publishing Institution:
- National Institute for Basic Biology Press Release
- Chuo University Press Release
- National Institute of Polar Research Press Release
- High Energy Accelerator Research Organization Press Release
- Tohoku University Press Release(Tohoku University Website, Laboratory Website)
- University of Hyogo Press Release